Heat Treatment Furnaces
Heat treating is a group of industrial and metalworking processes used to alter the physical, and sometimes chemical, properties of a material. The most common application is metallurgical. Heat treatments are also used in the manufacture of many other materials, such as glass. Heat treatment involves the use of heating or chiller, normally to extreme temperatures, to achieve a desired result such as hardening or softening of a material. Heat treatment techniques include annealing, case hardening, precipitation strengthening, tempering and quenching.


Heat Treatment Furnace by Micron has installed a brand new (2006) heat treatment plant capable of soaking temperatures up to 1050ºC. Featuring fully programmable cycles, the fully automatic electric furnace improves metallurgical control and optimises mechanical properties due to supreme heat uniformity. The working envelope of 1200 x 1200 x 2000mm caters for loads of up to 2 Tonnes.
Heat Treatment Techniques
It is noteworthy that while the termheat treatment applies only to processes where the heating and cooling are done for the specific purpose of altering properties intentionally, heating and cooling often occur incidentally during other manufacturing processes such as hot forming or welding. Heat treatment involves the use of heating or chilling, normally to extreme temperatures, to achieve a desired result such as hardening or softening of a material. Heat treatment techniques include annealing, case hardening, precipitation strengthening, tempering and quenching.
Metallic materials consist of a microstructure of small crystals called “grains” or crystallites. Heat treatment provides an efficient way to manipulate the properties of the metal by controlling the rate of diffusion and the rate of cooling within the microstructure. There are two mechanisms that may change an alloy’s properties during heat treatment.

Castings fresh from the heat treatment furnace

Heat Treatment Processing
The crystal structure consists of atoms that are grouped in a very specific arrangement, called a lattice. This rearrangement, called allotropy or polymorphism, may occur several times, at many different temperatures for a particular metal. When in the soluble state, the process of diffusion causes the atoms of the dissolved element to spread out, attempting to form a homogenous distribution within the crystals of the base metal.
If the alloy is cooled to an insoluble state, the atoms of the dissolved constituents (solutes) may migrate out of the solution. This type of diffusion, called precipitation, leads to nucleation, where the migrating atoms group together at the grain-boundaries. Unlike iron-based alloys, most heat treatable alloys do not experience a ferrite transformation.
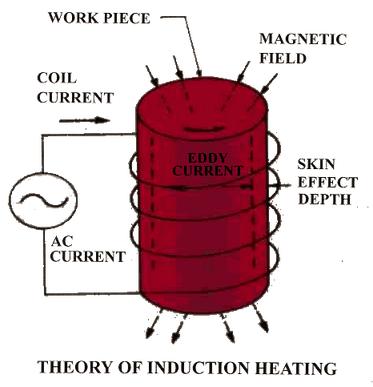
Theory of Induction Heating

Induction Heat Treatment

Quenching
Many metals and non-metals exhibit a martensite transformation when cooled quickly. When the crystal matrix changes to its low temperature arrangement, the atoms of the solute become trapped within the lattice.
When some alloys are cooled quickly, such as steel, the martensite transformation hardens the metal, while in others, like aluminum, the alloy becomes softer.










 Alloy Suppliers
Alloy Suppliers  Aluminum
Aluminum  Aluminum Extrusions
Aluminum Extrusions  Copper-Brass-Bronze
Copper-Brass-Bronze  Nickel
Nickel  Magnets
Magnets  Stainless Steel
Stainless Steel  Stainless Steel Tubing
Stainless Steel Tubing  Steel Service Centers
Steel Service Centers  Titanium
Titanium  Tungsten
Tungsten  Wire Rope
Wire Rope