Boron
Boron is a non metallic element and the only non-metal of the group 13 of the periodic table the elements. Boron is electron-deficient, possessing a vacant p-orbital. It has several forms, the most common of which is amorphous boron, a dark powder, unreactive to oxygen, water, acids and alkalis. It reacts with metals to form borides. At standard temperatures boron is a poor electrical conductor but is a good conductor at high temperatures.
 Boron
Boron
Boron is the chemical element with atomic number 5 and the chemical symbol B. Because boron is produced entirely by cosmic ray spallation and not by stellar nucleosynthesis, it is a low-abundance element in both the solar system and the Earth’s crust. However, boron is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. These are mined industrially as evaporate ores, such as borax and kernite.
You might also like
| Semiconductor Materials What is Semiconductor ? A semiconductor... | What is Ceramics ? Ceramics - A Definition A ceramic is an... | Properties of Metal Metal Properties - Overview A metal is a... | Silicon Wafer Silicon Wafer Manufacturing - How does it... |

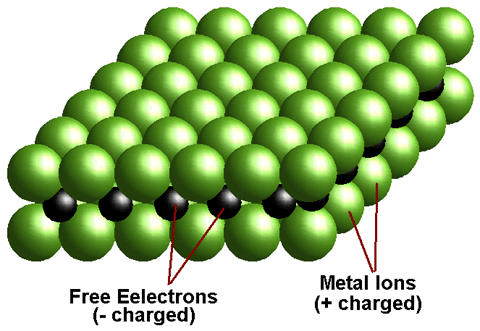
 Alloy Suppliers
Alloy Suppliers
 Aluminum
Aluminum
 Aluminum Extrusions
Aluminum Extrusions
 Copper-Brass-Bronze
Copper-Brass-Bronze
 Nickel
Nickel
 Magnets
Magnets
 Stainless Steel
Stainless Steel
 Stainless Steel Tubing
Stainless Steel Tubing
 Steel Service Centers
Steel Service Centers
 Titanium
Titanium
 Tungsten
Tungsten
 Wire Rope
Wire Rope