When a piece of a magnetic material is made smaller and smaller, is acquires simpler magnetic domain structure since less domain walls are needed in order to minimize the stray field energy. The extreme limit is represented by single domain particles. Magnetic nanoparticles are useful for a wide range of applications from data storage to medicinal imaging.
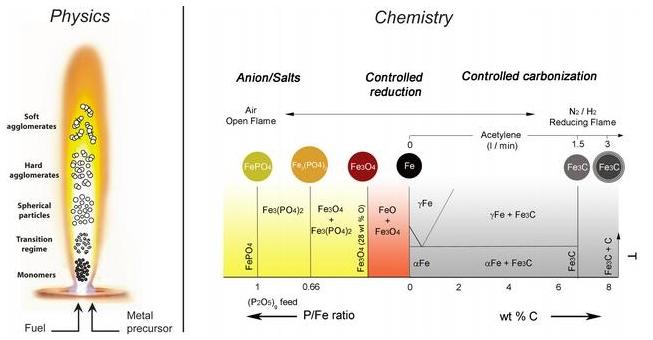 Various flame spray conditions and their impact on the resulting nanoparticles
Various flame spray conditions and their impact on the resulting nanoparticles
High capacity information storage, for instance, requires smaller particle size that decreasing the particle size lowers the anisotropy energy responsible for holding the magnetic moments along certain directions and it becomes comparable to the thermal energy. Thermal fluctuations randomize magentic moments (unless external magnetic field is applied), which is the essence of the so-called superparamagnetic behavior. Core/shell structured magnetic systems have an extra source of anisotropy that increases the stability of magnetic moments up to certain temperature called blocking temperature.
Magnetic nanoparticles are a class of nanoparticle which can be manipulated using magnetic field. Such particles commonly consist of magnetic elements such as iron, nickel and cobalt and their chemical compounds. The magnetic nanoparticles have been the focus of much research recently because they possess attractive properties which could see potential use in catalysis, biomedicine, magnetic resonance imaging,magnetic particle imaging, data storage and environmental remediation.
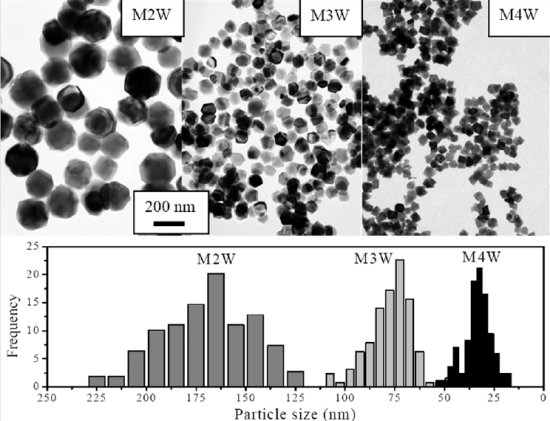 TEM images and histograms for magnetite nanoparticles synthesized at different Fe (II) concentrations
TEM images and histograms for magnetite nanoparticles synthesized at different Fe (II) concentrations
The physical and chemical properties of magnetic nanoparticles largely depend on the synthesis method and chemical structure. Current magnetic-nanoparticle technology is challenging due to the limited magnetic properties of iron oxide nanoparticles. Increasing the saturation magnetization of magnetic nanoparticles may permit more effective development of multifunctional agents for simultaneous targeted cell delivery, magnetic resonance imaging contrast enhancement, and targeted cancer therapy in the form of local hyperthermia. Delaware researchers have recently synthesized novel iron-based nanoparticles (FeNPs) coated with biocompatible bis-carboxyl-terminated polyethylene glycol.
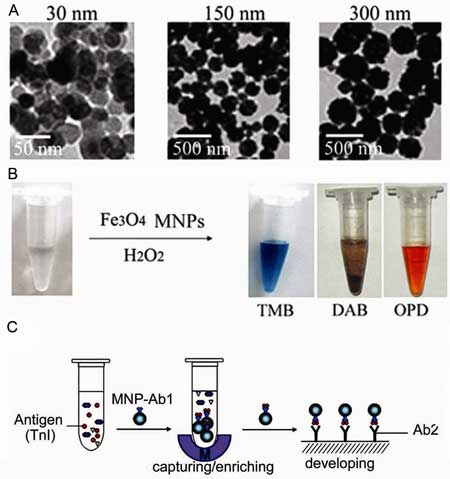 Fe3O4 MNPs show peroxidase-like activity. A) TEM images of Fe3O4 MNPs of different sizes. B) The Fe3O4 MNPs catalyze oxidation of various peroxidase substrates in the presence of H2O2 to produce different color reactions. C) Immunoassays based on the peroxidase activity of Fe3O4 magnetic nanoparticles. (Image: Prof. Yan/CAS)
Fe3O4 MNPs show peroxidase-like activity. A) TEM images of Fe3O4 MNPs of different sizes. B) The Fe3O4 MNPs catalyze oxidation of various peroxidase substrates in the presence of H2O2 to produce different color reactions. C) Immunoassays based on the peroxidase activity of Fe3O4 magnetic nanoparticles. (Image: Prof. Yan/CAS)
In comparison to conventional iron oxide nanoparticles similar in size (10 nm), FeNPs particles have a much greater magnetization and coercivity based on hysteresis loops from sample magnetometry. The last five years have witnessed an exponential growth in activities associated with the potential use of magnetic nanoparticles in biomedical applications. It is well known that size and composition influence the bio-application of magnetic nanoparticles.
Thus, for applications in angiography and tumour permeability, ultra-small superparamagnetic iron oxide particles are preferred. For hyperthermia treatment, particles with sizes around the monodomain–multidomain transition, i.e. particles below 50 nm in diameter, have been found to produce the maximum specific absorption rate (SAR). It has been reported that the SAR of 35 nm magnetite particles is twice that of 10nbsp;nm particles.
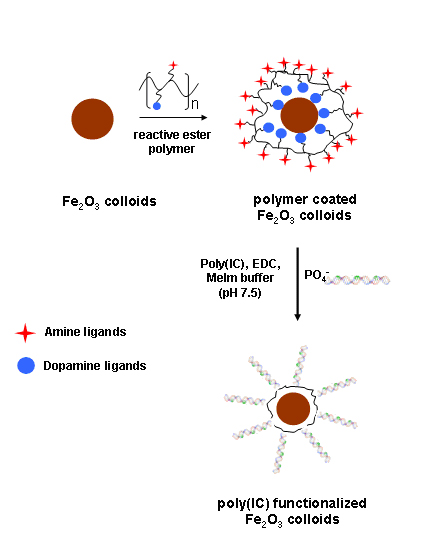
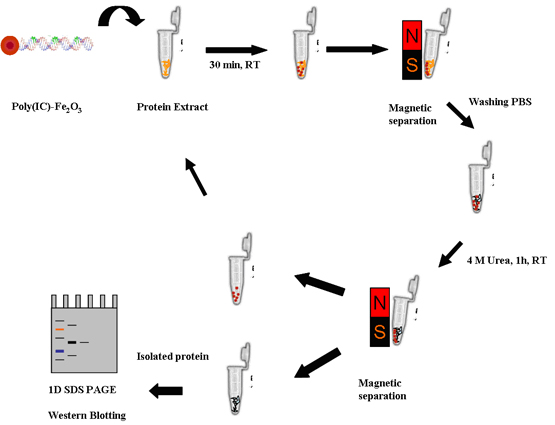 Functionalized Magnetic Nanoparticles at Protein Separation - Here polymer coated superparamagnetic gamma-Fe2O3 nanoparticles were derivatized with a synthetic double-stranded RNA [poly(IC)], a known allosteric activator of the latent (2-5)A synthetase, to separate a single 35 kDa protein from a crude extract which cross reacted with antibodies, raised against the sponge enzyme.
Functionalized Magnetic Nanoparticles at Protein Separation - Here polymer coated superparamagnetic gamma-Fe2O3 nanoparticles were derivatized with a synthetic double-stranded RNA [poly(IC)], a known allosteric activator of the latent (2-5)A synthetase, to separate a single 35 kDa protein from a crude extract which cross reacted with antibodies, raised against the sponge enzyme.
Specifically, the first section of this review describes synthetic routes producing equiaxial magnetic iron oxide particles below 10 nm which exhibit superparamagnetism combined with surface effects; those between 10 and 30 nm which exhibit superparamagnetic effects and some blocking, above 30 nm which are at the monodomain–multidomain boundary and also methods that produce anisometric nanoparticles.
Ferrite nanoparticles are the most explored magnetic nanoparticles up to date. Once the ferrite nanoparticles become smaller than 128 nm they become superparamagnetic which prevents self agglomeration since they exhibit their magnetic behavior only when an external magnetic field is applied. With the external magnetic field switched off, the remanence falls back to zero. Just like non-magnetic oxide nanoparticles, the surface of ferrite nanoparticles is often modified by surfactants, silicones or phosphoric acid derivatives to increase their stability in solution.
 Functionalized Magnetic Nanoparticles at Drug Transport
Functionalized Magnetic Nanoparticles at Drug Transport
Magnetic nanoparticles are used in an experimental cancer treatment called magnetic hyperthermia in which the fact that nanoparticles heat when they are placed in an alternative magnetic field is used.Another potential treatment of cancer includes attaching magnetic nanoparticles to free-floating cancer cells, allowing them to be captured and carried out of the body. Magnetic nanoparticles can be used for the detection of cancer.
Blood can be inserted onto a microfluidic chip with magnetic nanoparticles in it. These magnetic nanoparticles are trapped inside due to an externally applied magnetic field as the blood is free to flow through. The magnetic nanoparticles are coated with antibodies targeting cancer cells or proteins. The magnetic nanoparticles can be recovered and the attached cancer-associated molecules can be assayed to test for their existence.
Magnetic nanoparticles can be conjugated with carbohydrates and used for detection of bacteria. Magnetic immunoassay (MIA) is a novel type of diagnostic immunoassay utilizing magnetic beads as labels in lieu of conventional, enzymes , radioisotopes or fluorescent moieties. The presence of magnetic beads is then detected by a magnetic reader (magnetometer) which measures the magnetic field change induced by the beads. Magnetic nanoparticles are being used or have the potential use as a catalyst or catalyst supports.
Magnetic nanoparticles of, for example, magnetite or maghemite (common iron oxide materials used in different biomedical applications or in data storage systems) with diameters smaller than about 50 nm are singledomains. Magnetic nanoparticles can be divided into particles that are superparamagnetic or thermally blocked. Superparamagnetic particles have magnetic relaxation times that are shorter compared to the typical time scale of the measurement.
Thermally blocked particles have magnetic relaxation times that are larger when compared with a typical time scale of measurement being used to study the particle system. If the nanoparticles are placed in a solid matrix, the thermally blocked nanoparticles will exhibit both remanence and coercivity while the superparamagnetic particles will not show any remanence and coercivity. Coercivity is the field that brings the magnetization to zero value while remanance is the residual magnetization of the particle system after being magnetically saturated with an external magnetic field.
In chemistry, a catalyst support is the material, usually a solid with a high surface area, to which a catalyst is affixed. Magnetic CoPt nanoparticles are being used as an MRI contrast agent for transplanted neural stem cell detection. Grain sizes can be as small as 3 nanometers. Magnetic nanoparticles can be used for a variety of genetics applications. One application is the isolation of mRNA. Invitrogen is a supplier of such magnetic Dynabeads. Magnetic beads have also been used in plasmid assembly.
You might also like
| What is Nanoparticle ? Nanoparticles are particles that have one... | Austenite (Gamma Iron) Austenite, also known as gamma phase... | Silver Nanoparticle Silver nanoparticles are nanoparticles of... | Whai is Stainless Steel? A Stainless Steel art deco sculpture... |



 Alloy Suppliers
Alloy Suppliers
 Aluminum
Aluminum
 Aluminum Extrusions
Aluminum Extrusions
 Copper-Brass-Bronze
Copper-Brass-Bronze
 Nickel
Nickel
 Magnets
Magnets
 Stainless Steel
Stainless Steel
 Stainless Steel Tubing
Stainless Steel Tubing
 Steel Service Centers
Steel Service Centers
 Titanium
Titanium
 Tungsten
Tungsten
 Wire Rope
Wire Rope